Industry Solutions:
From Lab-Scale Research to Clinical Reality
Providing specialized technical advisory in Soft Robotics, Bio-Digital Twins, and MedTech Validation for global healthcare innovators.
Diagnostic Automation
Robotic ultrasound
Autonomous robotic ultrasound (RUS) for standardized imaging.
Accelerating R&D for AI-driven and teleoperated diagnostic platforms. Also, performing ethical auditing for Healthcare AI.
Navigational Dynamics (PyChrono)
PyChrono autonomous navigation model for a flexible endoscope.
Real-time Multi-body collision avoidance in constrained anatomical environments.
We reduce MedTech R&D cycles through high-fidelity virtual prototyping and physics-based digital twins (in SOFA or PyChrono) to identify mechanical bottlenecks before you commit to hardware.






Medical Device Regulation and Commercialization (Lap-Pack)
Lap-Pack is a low fidelity laparoscopic trainer for early and experienced clinical trainees - created by Dr. Manish Chauhan.
We are capable of bringing lab based prototypes to industry ready products.
Expert in evaluating clinical instrumentation safety and providing the technical documentation required for regulatory compliance (Targeting IEC 60601-1 Compliance).
We bridge the gap for international MedTech firms by aligning product capabilities with UK National Standards, ISO requirements, and the Gatsby Benchmarks for educational robotics.
The Challenge
Detecting paediatric pneumonia in low-resource settings is hindered by a lack of expert radiologists and high inter-observer variability in X-ray interpretation. Most AI solutions (CNNs) are "black boxes" that lack the transparency required for clinical trust and regulatory audit.
Selective Case Study
The Solution
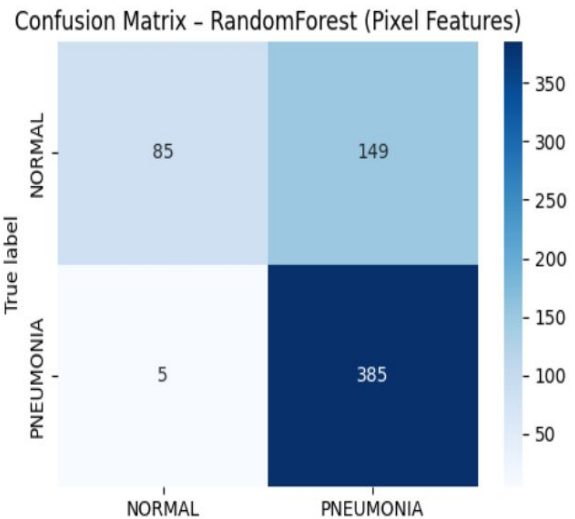
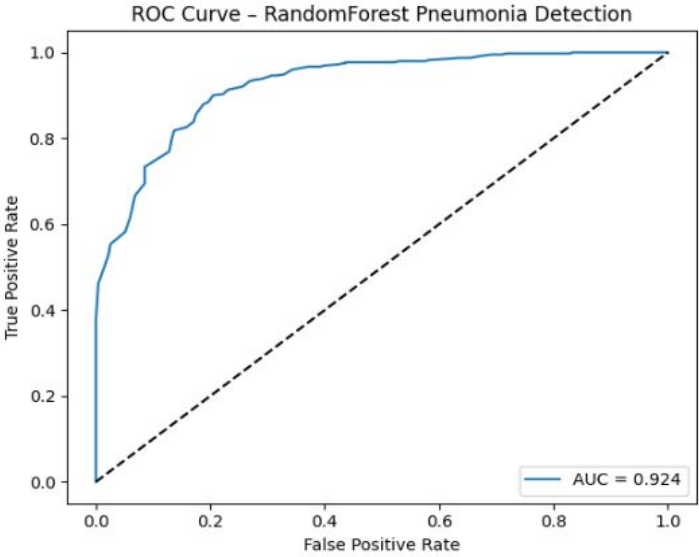
I led the development of a Random Forest Classification Framework using a dataset of 5,800+ paediatric chest X-rays. Unlike deep learning models, this ensemble approach was chosen to prioritize clinical interpretability and computational efficiency, allowing for deployment on low-spec hardware in remote clinics.
The Technical Impact
Safety-Critical Optimization: Achieved a 98.7% Recall (Sensitivity) for pneumonia detection, intentionally minimizing false negatives to ensure no life-threatening cases were missed.
Data Integrity and Auditing: Implemented a robust Traceability Pipeline using independent training/test splits to prevent data leakage—meeting industrial standards for AI validation.
Clinical Logic: Demonstrated a transparent diagnostic baseline that identifies pathological patterns (opacities) with higher consistency than manual triage in high-volume environments.
Aligned with SaMD (Software as a Medical Device) Validation protocols.
In delivering these projects, I apply industry-standard methodologies including:
Risk Management: ISO 14971 (Hazard Analysis & Risk Mitigation).
Validation: Verification & Validation (V&V) protocols for SaMD (Software as a Medical Device).
Quality: Data integrity aligned with ALCOA+ principles for clinical AI training.
Technical Insight - For key industry solutions
Contact
Reach out for collaborations or questions.
Phone
manish.chauhan[at]york.ac.uk
+44 (0) 1904 32 2361
© 2025. All rights reserved.
